Pesticides are compounds that are used to kill pests, including insects, rodents, fungi and unwanted plants (e.g. weeds). When pesticides are used to protect crops, their residues will potentially affect and threaten human and environmental health. For that reason, they must be monitored and used safely, or even avoided, if possible.
In order to see the clear picture of the potential harm of pesticides, let’s take dichlorodiphenyltrichloroethane, commonly known as DDT, for example.
A Brief History of DDT
DDT is an organochlorine pesticide which was used as an all-purpose insecticide. Before the introduction of DDT, insect-borne diseases like malaria, typhus, yellow fever, and others killed millions of people worldwide. As the use of DDT spread over time, some scientists noticed that its incautious use was doing more harm than good to populations worldwide.
Once it enters the organism, DDT is metabolized and excreted more slowly than the nutrients that are passed from one trophic level to the next. That way DDT accumulates in the bodies, especially in fat tissue. Therefore, the biomagnification process occurs and the harmful effects of DDT hit
In 2004, the Stockholm Convention on Persistent Organic Pollutants, which was signed by 170 countries, restricted the use of DDT to emergency insect control, meaning that it could be used only in the events of certain insect-borne diseases outbreaks (e.g. malaria). However, in some countries, DDT is still used regularly for controlling mosquitos and other insects.
Methods for Pesticide Detection
There are multiple methods used for the detection of pesticides, such as fluorescence spectrophotometry, gas chromatography with mass spectrometry detection, high-performance liquid chromatography (HPLC) with fluorescence detection, and HPLC coupled to mass spectrometry. In contrast, electrochemical methods are cheap, sensitive, selective and also quite undemanding.
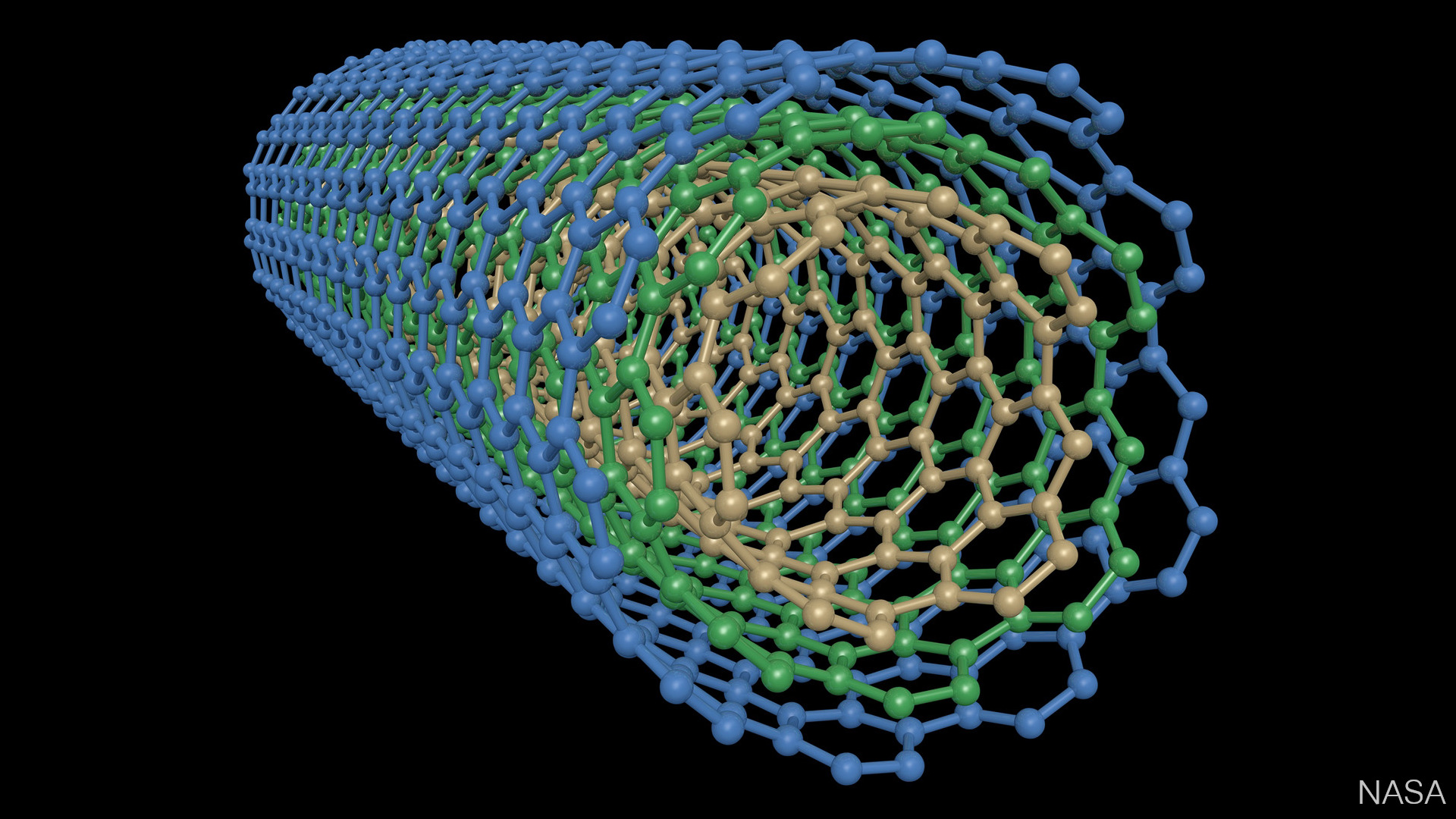
Carbon nanotubes have provided outstanding advances in the design of electrochemical sensors. Multi-walled carbon nanotubes are composed of sp2 carbon units positioned as multiple concentric tubes of rolled-up graphene. Numerous advantages like remarkable active surface area, high strength, chemical inertness, and low charge-transfer resistance in aqueous solutions make carbon nanotubes convenient for electrochemical analysis


Hello There. I found your blog using msn. This is a very well written article. I’ll be sure to bookmark it and return to read more of your useful info. Thanks for the post. I will certainly return.
Pretty! This was an incredibly wonderful post. Thanks for supplying this information.
Usually I do not learn article on blogs, however I would like to say that this write-up very forced me to take a look at and do it! Your writing style has been amazed me. Thank you, quite nice post.
Wonderful blog! Do you have any helpful hints for aspiring writers? I’m hoping to start my own blog soon but I’m a little lost on everything. Would you advise starting with a free platform like WordPress or go for a paid option? There are so many options out there that I’m totally overwhelmed .. Any suggestions? Cheers!
Your post extremely cool. I glad to be here. I enjoyed reading your articles. Thanks and keep up the good work!