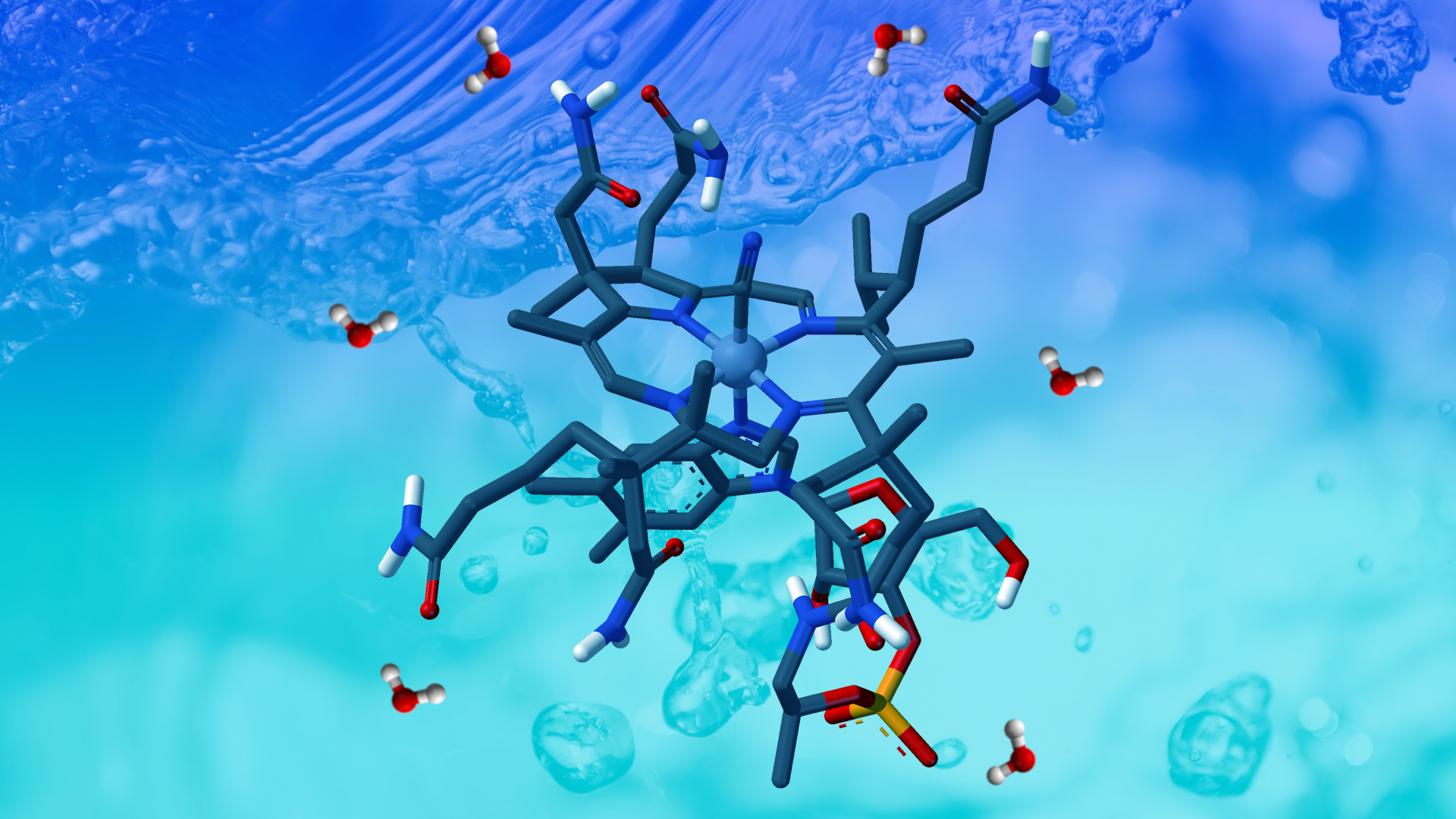
Every living organism on Earth uses coenzymes in various important enzyme-catalyzed reactions, for example, ones that are involved in the central metabolic pathways such as glycolysis and the Krebs cycle. Many species can synthesize their own coenzymes from simple precursors, which is very important in four out of five kingdoms: prokaryotes, protozoa, fungi, and plants. On the other hand, animals don’t have the ability to produce some of the coenzymes, hence they have to consume it through their diet.
Depending on the interaction with apoenzymes, two types of coenzymes are known – cosubstrates and prosthetic groups. Cosubstrates are actually substrates in the enzyme-catalyzed reactions. They are altered throughout the biochemical reaction and dissociate from the active site. Oppositely, the prosthetic group remains bonded for the enzyme during the course of the reaction. Depending on the nature of the chemical bonds in the active site, a prosthetic group can form a strong covalent bond with its apoenzyme or it can be weakly bound to the active center by many weak interactions. Similarly to ionic amino acid residues of the active site, the prosthetic group must return to its original form by the end of the catalytic event or the holoenzyme won’t be catalytically active anymore.
As mentioned earlier, animals lost their ability to synthesize some coenzymes. Mammals—including humans—have the ability to source necessary coenzymes or their direct precursors in order to survive. Herbivores obtain vitamins from plants and microorganisms, and carnivores get their vitamins from meat. Majority of consumed vitamins are enzymatically converted into their corresponding coenzymes.
Vitamins soluble in water are needed in small quantities because cell depots of their coenzymes are unstable. Therefore, the excess of these type of vitamins is excreted via urine. On the other side, lipid vitamins such as A, D, E, I and K are stored in animals’ bodies and increased intake can cause hypervitaminoses, because—unlike hydrosoluble vitamins—the excess can’t be excreted that easy.
The story continues in the review paper Mechanisms of actions of coenzymes published in our journal (CN: 1(1), 153-183), in which Dr Biljana Arsic, a researcher at the Department of Chemistry in Nis and member of the Royal Society of Chemistry, describes nucleotide co-substrates, NAD+ and NADP+, FAD and FMN, coenzyme A, thiamine pyrophosphate, pyridoxal phosphate, biotin, tetrahydrofolate, cobalamin, lipoamide, ubiquinone, protein coenzymes and cytochromes, as well as their behavior in living organisms.

Someone essentially lend a hand to make significantly articles I would state. This is the first time I frequented your website page and thus far? I amazed with the analysis you made to make this actual post extraordinary. Fantastic activity!
Excellent goods from you, man. I have understand your stuff previous to and you’re just too excellent. I actually like what you have acquired here, certainly like what you’re stating and the way in which you say it. You make it enjoyable and you still take care of to keep it sensible. I can not wait to read far more from you. This is really a terrific web site.
Hi there! This post couldn’t be written much better!
Reading through this article reminds me of my previous roommate!
He constantly kept preaching about this. I am going to forward this post to him.
Pretty sure he’s going to have a great read. Many thanks for sharing!
Greate post. Keep writing such kind of information on your page.
Im really impressed by your blog.
Hello there, You have performed an incredible job.
I’ll definitely digg it and personally recommend to my friends.
I’m sure they’ll be benefited from this website.
Keep on working, great job!
I get pleasure from, cause I found exactly what I used to be having a look for.
I truly appreciate this blog post.Really looking forward to read more. Really Great.
I just want to say I’m newbie to blogging and site-building and absolutely savored your web page. Likely I’m planning to bookmark your website . You definitely have exceptional stories. Thanks for revealing your blog.
I simply want to tell you that I am just all new to blogs and certainly enjoyed this website. Very likely I’m likely to bookmark your blog post . You certainly come with amazing posts. Thank you for sharing with us your website page.
I was able to find good info from your blog posts.
That is a really good tip especially to those fresh to the blogosphere. Brief but very precise info… Thank you for sharing this one. A must read post!
You’re so cool! I do not believe I’ve truly read something like this before. So nice to discover somebody with a few genuine thoughts on this subject matter. Seriously.. thank you for starting this up. This web site is something that’s needed on the internet, someone with some originality!
Very good post! We are linking to this great article on our site. Keep up the good writing.
Your style is really unique compared to other folks I’ve read stuff from. Thank you for posting when you’ve got the opportunity, Guess I will just bookmark this site.
Saved as a favorite, I really like your web site!
Spot on with this write-up, I seriously feel this amazing site needs a great deal more attention. I’ll probably be back again to read more, thanks for the information!
Thank you for your article. Really Cool.
You’re so awesome! I do not think I’ve truly read anything like that before. So nice to find someone with a few genuine thoughts on this subject. Really.. thank you for starting this up. This site is one thing that is needed on the internet, someone with a bit of originality!
Pretty! This has been an incredibly wonderful article. Thanks for supplying these details.
I need to to thank you for this great read!! I absolutely loved every bit of it. I have you saved as a favorite to check out new stuff you post…
Having read this I thought it was really enlightening. I appreciate you finding the time and energy to put this article together. I once again find myself spending a lot of time both reading and posting comments. But so what, it was still worth it!
I was very happy to uncover this website. I want to to thank you for your time for this particularly wonderful read!! I definitely appreciated every bit of it and i also have you book-marked to see new information in your site.
Your style is very unique in comparison to other people I’ve read stuff from. Thank you for posting when you’ve got the opportunity, Guess I will just book mark this web site.
After exploring a number of the blog posts on your site, I honestly like your way of blogging. I saved as a favorite it to my bookmark webpage list and will be checking back soon. Please visit my website too and let me know what you think.
I wanted to thank you for this great read!! I certainly loved every bit of it. I have got you saved as a favorite to look at new stuff you post…
I was able to find good information from your blog articles.
Wonderful article! We are linking to this particularly great post on our site. Keep up the great writing.
There’s definately a lot to learn about this subject. I love all of the points you made.
I’m extremely pleased to discover this site. I need to to thank you for your time just for this fantastic read!! I definitely appreciated every bit of it and i also have you book marked to check out new information on your website.
Way cool! Some very valid points! I appreciate you writing this post and also the rest of the site is really good.
I was excited to discover this great site. I want to to thank you for ones time for this particularly wonderful read!! I definitely loved every little bit of it and i also have you book-marked to see new stuff in your site.
I used to be able to find good info from your content.
I’m impressed, I have to admit. Seldom do I encounter a blog that’s both educative and interesting, and without a doubt, you’ve hit the nail on the head. The problem is something not enough people are speaking intelligently about. I’m very happy I found this in my hunt for something regarding this.
Great post! We will be linking to this great post on our website. Keep up the good writing.
Your style is really unique compared to other people I’ve read stuff from. Many thanks for posting when you’ve got the opportunity, Guess I’ll just bookmark this site.
Very nice article. I certainly appreciate this site. Continue the good work!
This blog was… how do you say it? Relevant!! Finally I’ve found something which helped me. Thanks!
You should take part in a contest for one of the most useful blogs on the net. I will recommend this site!
I’m excited to uncover this site. I want to to thank you for your time for this particularly wonderful read!! I definitely enjoyed every little bit of it and I have you bookmarked to check out new things on your web site.
Good day! I simply want to give you a huge thumbs up for your excellent information you have got right here on this post. I am returning to your blog for more soon.
Spot on with this write-up, I honestly feel this website needs a great deal more attention. I’ll probably be back again to read more, thanks for the info!
It’s hard to find well-informed people for this topic, but you sound like you know what you’re talking about! Thanks
I wanted to thank you for this fantastic read!! I certainly enjoyed every little bit of it. I’ve got you book-marked to check out new things you post…
Excellent site you have got here.. It’s hard to find high-quality writing like yours these days. I seriously appreciate people like you! Take care!!
I could not resist commenting. Exceptionally well written!
Wow, fantastic blog layout! How long have you been blogging for? you made blogging look easy. The overall look of your web site is great, as well as the content!
You need to take part in a contest for one of the greatest sites on the web. I will highly recommend this site!
I blog often and I seriously appreciate your content.
Your article has truly peaked my interest. I will take a note
of your blog and keep checking for new information about once a week.
I opted in for your Feed too.
Generally I don’t learn article on blogs, however I wish to say that this write-up forced me to try. Your writing taste is amazing. Thank you, very nice article.
wow, awesome article post.Thanks Again. Will read on…
Hi my friend! I want to say that this post is awesome, nice written and include approximately all important infos. I’d like to see more posts like this .
I simply want to tell you that I am new to blogs and really loved this blog. More than likely I’m want to bookmark your blog . You definitely come with remarkable stories. Thank you for revealing your web page.
We absolutely love your blog and find nearly all of your post’s to be exactly what I’m looking for. Would you offer guest writers to write content available for you? I wouldn’t mind publishing a post or elaborating on a few of the subjects you write about here. Again, awesome site!
Very nice post. I just stumbled upon your weblog and wanted to say that I’ve truly enjoyed surfing around your blog posts. In any case I will be subscribing to your feed and I hope you write again soon!
Hello there, I found your blog via Google while looking for a related topic, your website came up, it looks great. I’ve bookmarked it in my google bookmarks.
Usually I don’t read article on blogs, but I wish to say that this write-up very forced me to try and do it! Your writing style has been amazed me. Thanks, quite nice article.
Good job on this article! I really like how you presented your facts and how you made it interesting and easy to understand. Thank you.
Heya this is a excellent write-up. I’m going to email this to my associates. I came on this while searching on aol I’ll be sure to come back. thanks for sharing.
I like what you guys are up too. Such clever work and reporting! Carry on the superb works guys I have incorporated you guys to my blogroll. I think it will improve the value of my website :).
Everything is very open with a very clear description of the issues. It was really informative. Your website is very useful. Many thanks for sharing!
I was excited to find this web site. I need to to thank you for ones time just for this fantastic read!! I definitely liked every bit of it and I have you bookmarked to look at new things on your blog.
I need to to thank you for this fantastic read!! I certainly loved every little bit of it. I’ve got you bookmarked to look at new things you postÖ
Hey, not necessarily you too great? Your own personal writing widens my know-how. Appreciate you.
I really liked this article. Thank you for sharing it. I’ll return to see more.
Hi there to all, the contents present at this web site are in fact awesome for people knowledge, well, keep up the nice work fellows.|
Great article.
I was excited to discover this page. I wanted to thank you for ones time due to this wonderful read!! I definitely enjoyed every little bit of it and I have you saved to fav to see new information on your site.
Having read this I believed it was rather enlightening. I appreciate you finding the time and energy to put this information together. I once again find myself personally spending a lot of time both reading and leaving comments. But so what, it was still worth it!
Thanks for the marvelous posting! I actually enjoyed reading it, you may be a great author.I will remember to bookmark your blog and definitely will come back sometime soon. I want to encourage you to continue your great job, have a nice afternoon!
Very nice blog post. I definitely appreciate this website. Keep writing!
I love what you guys tend to be up too. Such clever work and coverage! Keep up the superb works guys I’ve incorporated you guys to my own blogroll.|
Highly energetic article, I enjoyed that a lot. Will there be a part 2?|
Exactly the sort of content that enhances the web. Thanks!
I love the way you have actually ended this article …
Excellent site you have got here.. Itís hard to find high quality writing like yours these days. I truly appreciate individuals like you! Take care!!
If some one wishes expert view regarding blogging then i propose him/her to visit this webpage, Keep
up the pleasant work.
You are so interesting! I don’t suppose I have read anything like this before. So nice to discover somebody with a few original thoughts on this subject matter. Really.. thanks for starting this up. This site is something that’s needed on the internet, someone with a little originality!
I think that is among the most vital info for me. And i’m glad reading your article. However should observation on some common things, The site taste is wonderful, the articles is actually great : D. Just right activity, cheers|
Great write-up, I’m regular visitor of one’s blog, maintain up the nice operate, and It’s going to be a regular visitor for a lengthy time.
I could not resist commenting. Very well written!
Very good written article. It will be useful to anyone who employess it, as well as myself. Keep up the good work – for sure i will check out more posts.
I do not even know how I ended up here, but I thought this post was good. I do not know who you are but definitely you’re going to a famous blogger if you aren’t already 😉 Cheers!
You need to take part in a contest for top-of-the-line blogs on the web. I’ll recommend this website!
I think this is one of the most vital information for me. And i’m glad reading your article. But want to remark on few general things, The web site style is wonderful, the articles is really excellent : D. Good job, cheers
Just bookmarked this blog post as I have actually found it quite practical.
I have been browsing online greater than three hours nowadays, but I never discovered any fascinating article like yours. It’s beautiful worth enough for me. In my opinion, if all web owners and bloggers made just right content material as you did, the internet will be much more useful than ever before.
You are so great thanks
Great site you’ve got here.. Itís difficult to find high quality writing like yours these days. I really appreciate individuals like you! Take care!!
Your style is very unique compared to other folks I’ve read stuff from. Thank you for posting when you’ve got the opportunity, Guess I’ll just bookmark this web site.
This is a great tip especially to those new to the blogosphere. Short but very accurate info… Thanks for sharing this one. A must read article!
Hi! I just want to give you a huge thumbs up for your great information you have here on this post. I’ll be coming back to your website for more soon.
You need to take part in a contest for one of the finest sites on the internet. I am going to highly recommend this blog!
I have been exploring for a little bit for any high quality articles or blog posts on this sort of area . Exploring in Yahoo I at last stumbled upon this site. Reading this information So i’m happy to convey that I’ve a very good uncanny feeling I discovered exactly what I needed. I most certainly will make certain to don’t forget this web site and give it a glance regularly.
Excellent article! We will be linking to this great article on our website. Keep up the good writing.
You’re so interesting! I do not believe I’ve read a single thing like this before. So wonderful to find someone with a few original thoughts on this subject matter. Seriously.. thanks for starting this up. This site is something that is required on the internet, someone with some originality!
Pretty! This has been an extremely wonderful article. Many thanks for supplying this info.
This blog was… how do I say it? Relevant!! Finally I’ve found something that helped me. Many thanks!
You need to take part in a contest for one of the highest quality websites on the web. I am going to highly recommend this website!
Great site you have here.. It’s difficult to find quality writing like yours these days. I honestly appreciate people like you! Take care!!
Your style is so unique in comparison to other folks I’ve read stuff from. Many thanks for posting when you have the opportunity, Guess I’ll just book mark this blog.
Generally I do not read post on blogs, but I wish to say that this write-up very pressured me to check out and do it! Your writing style has been amazed me. Thank you, very great post.|
Excellent post. I definitely appreciate this site. Keep it up!
Your style is very unique compared to other people I’ve read stuff from. Thanks for posting when you have the opportunity, Guess I will just book mark this site.